Edit Studies
Explore Job Aids and Videos for more help:
Job Aids
Videos
To edit studies, follow the steps below:
- Click on the check box next to the name of the study.
- Click on the Edit button from the top main bar.
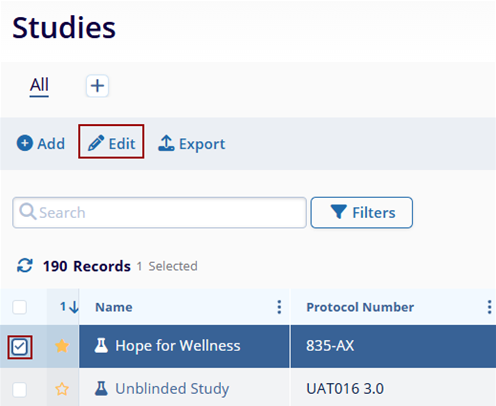
- Make any necessary changes to editable data within the Study Details by expanding the
following sections.General Information:
- Study Name*
- Primary Program
- Virtual Study
- Study Number
- Additional Program(s)
- Products
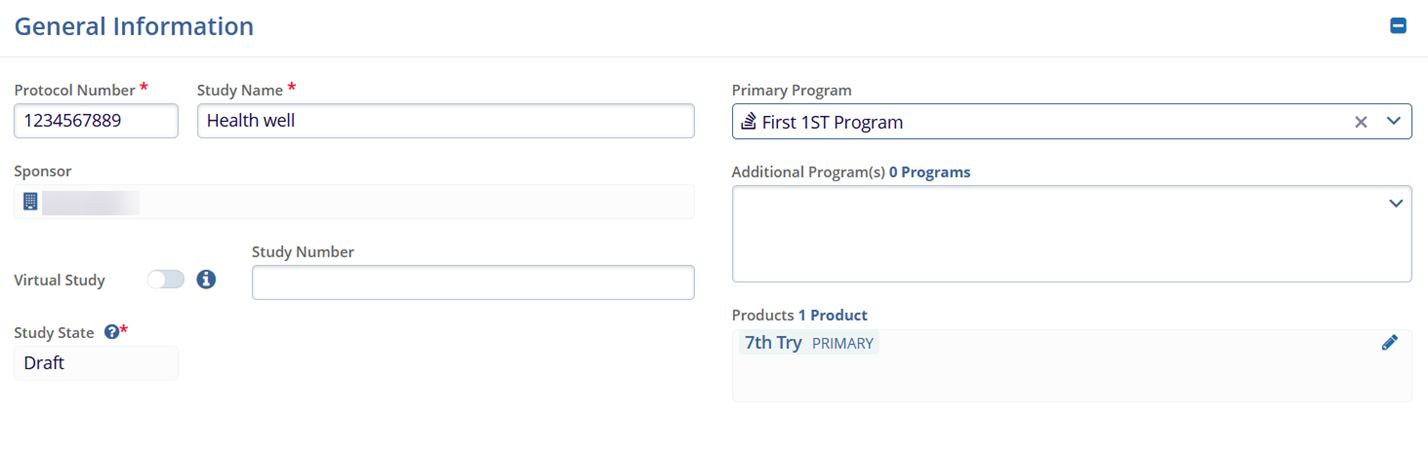
Protocol Information- Protocol Title*
- Protocol Summary
- Therapeutic Areas
- Indications
- Phase*
- Types
- Mechanisms
- Routes
- Objectives
- Designs*
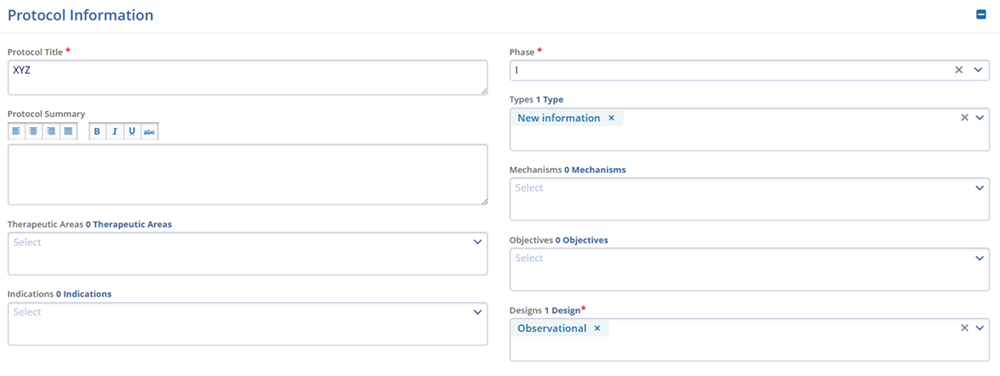
Project Management- Status
- Status Date
- Status Notes
- Projected Start Date
- Actual Start Date
- Projected End Date
- Actual End Date
- Database Lock Date
- Recruitment Months
- Site Recruitment Deadline
- Subject Recruitment Deadline
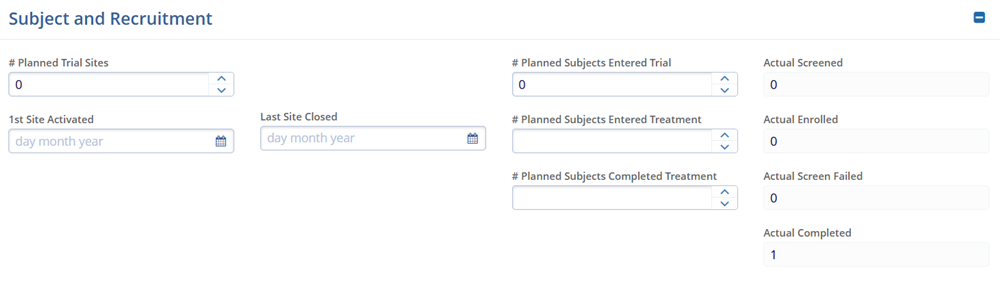
Subject and Recruitment- # Planned Trial Sites
- 1st Site Activated
- Last Site Closed
- # Planned Subjects Entered Trial
- # Planned Subjects Entered Treatment
- # Planned Subjects Completed Treatment
Informed Consent
Add Informed Consent
To add informed consent, follow the steps below:
- Click on the +Add button within the Informed Consent section.
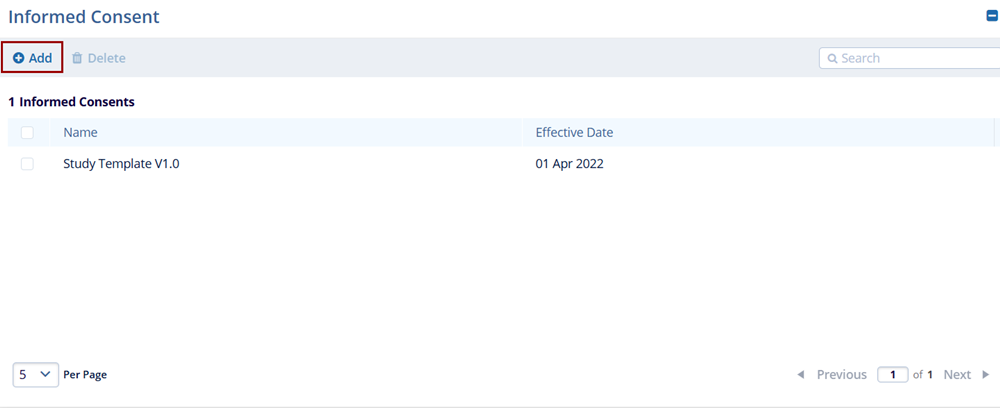
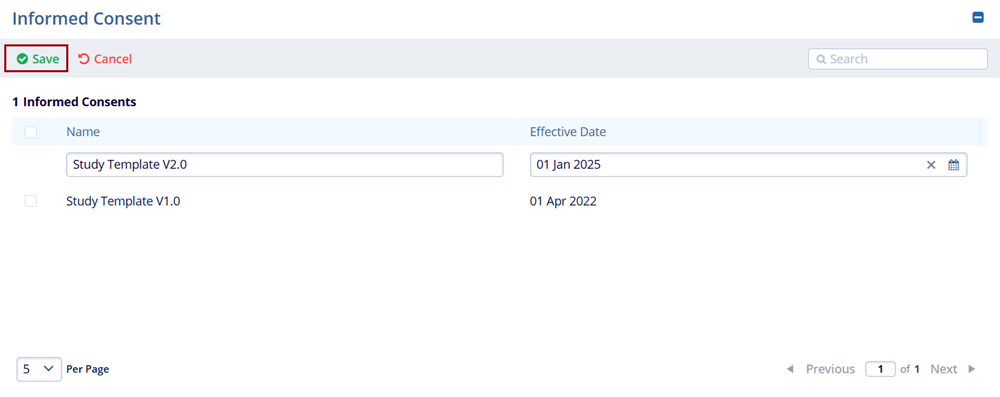
- Enter the Name and Effective Date into the respective fields.
- Click on the Save button.
Delete Informed Consent
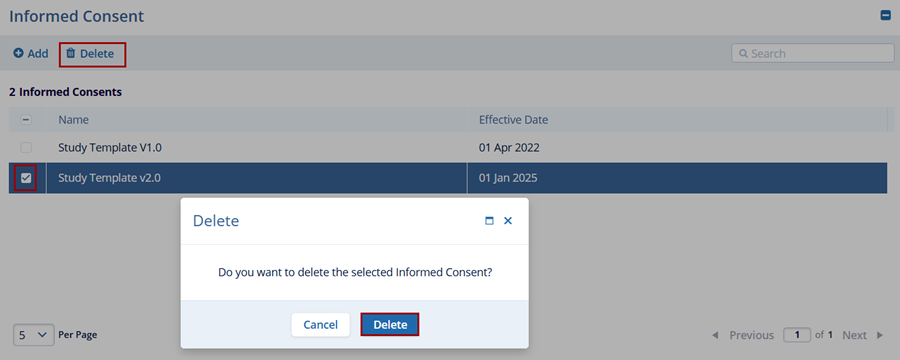
To delete an informed consent, follow the steps below:
- Select a record from the Informed Consent section to delete
- Click on the Delete button.
- On the confirmation popup, click on the Delete button.Note:
- ICFs can only be deleted when they are not linked to any other entity (e.g., Country, Site, Subject).
- For ICFs already in use, submit a Service Desk ticket for any data corrections.
Additional Information
The Additional Information section displays the custom fields configured via Settings > Fields > Studies. The displayed fields vary based on the configurations set in the Settings section for each customer.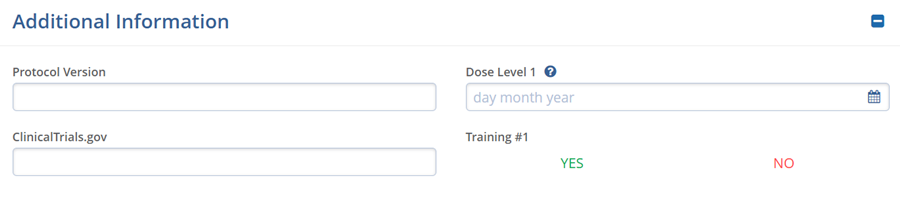
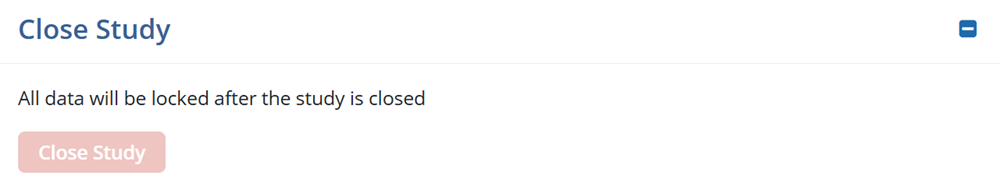
Close Study
The Close Study Section enables users to close a study when it is at the end of the life cycle.
- Click the Save button for the changes to apply.
- The Study Health Summary window is displayed showing items that are reconciled or open.
-
Navigate to the particular record that is open by clicking on the icon before the entity name and close the entity.
- Once all the related items are closed, the Yes, Close & Lock button is activated.
- Click the Yes, Close & Lock button.

- On the Close Study window, select the status date and click Close.

- Click the Save button for the changes to apply.
Didn’t find what you need?
Our dedicated Project Management and Client Services team will be available to meet your needs
24 hours a day, 7 days a week.
© 2025 Trial Interactive. All Rights Reserved


 Linkedin
Linkedin
 X
X

